37 lewis diagram for co2
Click here to get an answer to your question ✍️ Draw the Lewis dot structure of CO2 molecule. Lewis structure of CO2 has dot electron representative structure. Valence electrons of atoms undergo orbitals mixing in the chemical reactions, gives new types ...
10 May 2014 · 1 answerYou follow a sequence of steps. Explanation: Here are the steps that I follow when drawing a Lewis structure. 1. Decide which is the central ...
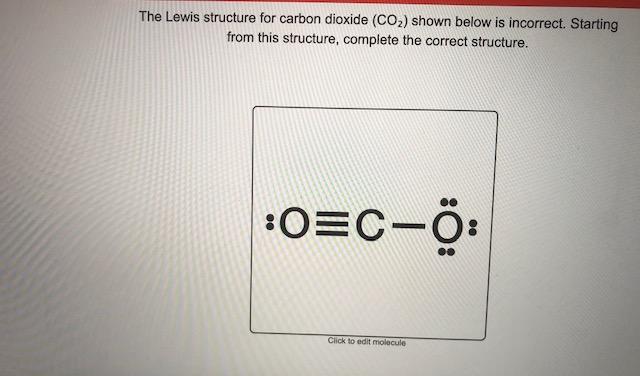
Lewis diagram for co2
In the lewis structure of CO 2, you can see there are two double bonds around carbon atom. Each oxygen atom has two lone pairs and carbon atom does not have a lone pair. Also, there are no charges in oxygen atoms and carbon atom. I quickly take you through how to draw the Lewis Structure of CO2 (Carbon DiOxide). I also go over hybridization, shape and bond angles. For Lewis structure of CO2, you will now have two Oxygen atoms forming double bonds with a Carbon atom. As all the valence electrons of all the atoms are used, there are no lone pairs of electrons or non-bonding pairs of electrons in the molecule.
Lewis diagram for co2. The first thing about the CO 2 Lewis structure is to put carbon in the center. Make both O atoms connect to C. There won’t be any bonds between the O’s directly. As a rule, carbon is always going to be in the center, and the other atoms connect to it. Second, connect the lone dots on each O to the C in the center. Each O needs to bond twice. The Lewis Dot Structure for carbon dioxide can be represented like this: o=C=o But what exactly does this mean? What is a Lewis Dot Structure, and what do the symbols in carbon dioxide’s structure represent? Let’s go over the Lewis structure and find out how to interpret this representation of carbon dioxide. How To Read A Lewis Dot Structure CO2 lewis structure contains two oxygen atoms and one carbon atom, connected with the double bond whereas carbon is the central atom, and no lone pair is present on it. But each oxygen in the CO2 lewis dot structure has two lone pairs. A lewis diagram helps us to know how electrons are arranged around individual atoms in a molecule. 6 Steps on How to Draw CO2’s Lewis Structure Calculate the total valence electrons found in a molecule. Carbon Valence Electron=4 Oxygen Valence electrons: 6*2 = 12 Total number of valence electrons = 16 Find the central atom, which is usually the one with the highest bonding sites, is the Carbon atom. Draw four dots around it.
Before we discuss the CO 2 lewis structure or lewis dot structure for CO2, we need to know the basics of lewis dot structure.Lewis dot structure work on the octet rule, which means that all the atoms in the structure would have eight electrons in their valence shell except hydrogen. Here is the Lewis structure of CO 2. CO2 Lewis structure. By following the above steps, you can draw the Lewis structure of any molecule. Here are some examples. Try to apply these five steps to the following examples and check whether your success or not. CO Lewis structure Step 01: calculation of total valence electrons of CO Step 02: CO2 Lewis Structure. The lewis structure of CO2 can be with some simple steps, but before that, it is important to understand lewis structure properly. So lewis structure generally gives us an idea about the nature of bonding and octet fulfillment of the atoms. According to the octet rule, an atom attains stability by fulfilling its octet. For Lewis structure of CO2, you will now have two Oxygen atoms forming double bonds with a Carbon atom. As all the valence electrons of all the atoms are used, there are no lone pairs of electrons or non-bonding pairs of electrons in the molecule.
I quickly take you through how to draw the Lewis Structure of CO2 (Carbon DiOxide). I also go over hybridization, shape and bond angles. In the lewis structure of CO 2, you can see there are two double bonds around carbon atom. Each oxygen atom has two lone pairs and carbon atom does not have a lone pair. Also, there are no charges in oxygen atoms and carbon atom.

Caption reads, "[John Lewis speaking at a meeting of American Society of Newspaper Editors, Statler Hilton Hotel, Washington, D.C.] / [MST]." Original black and white negative by Marion S. Trikosko. Taken April 16th, 1964, Washington D.C, United States (@libraryofcongress). Colorized by Jordan J. Lloyd. Library of Congress Prints and Photographs Division Washington, D.C. 20540 https://www.loc.gov/item/2003688130/



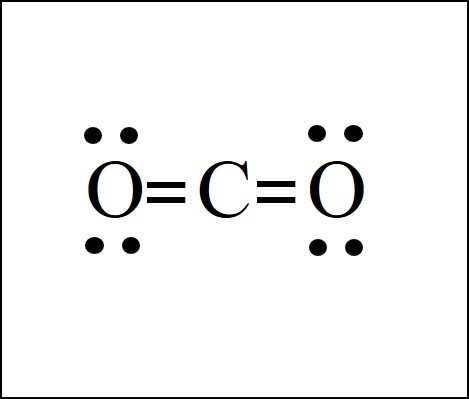


/CO2-Resonance-56a12a2b5f9b58b7d0bca8a3.png)





:max_bytes(150000):strip_icc()/CO2LewisStructure-591c94063df78cf5fadfde77.png)





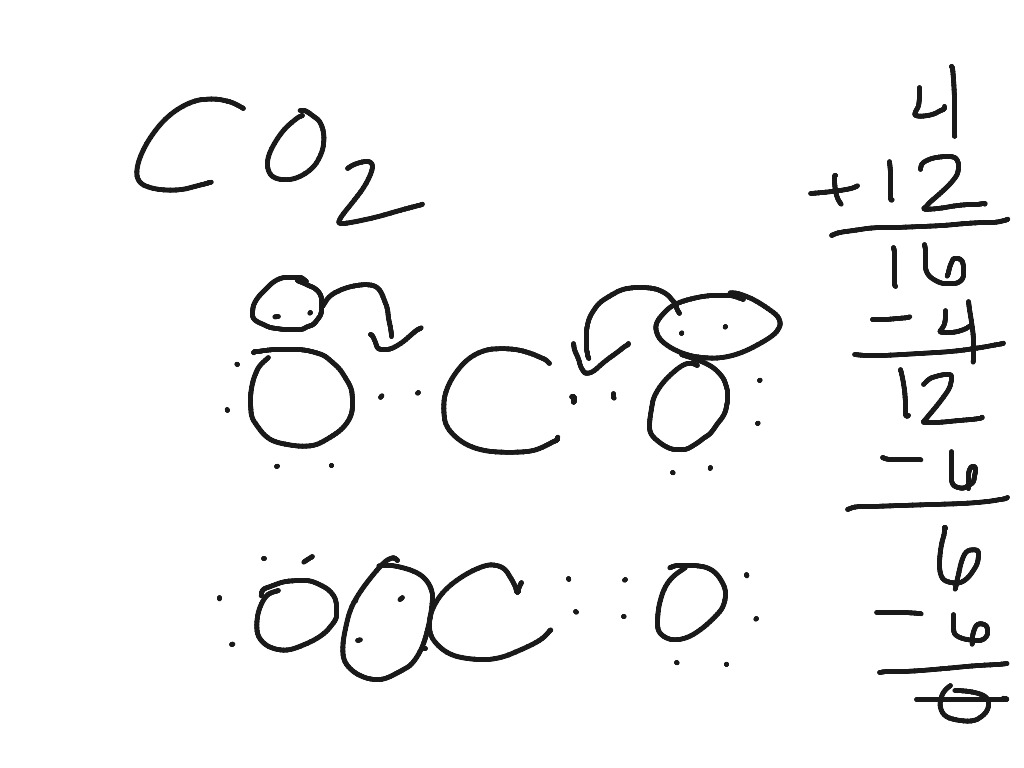


0 Response to "37 lewis diagram for co2"
Post a Comment