41 outer electron box diagram
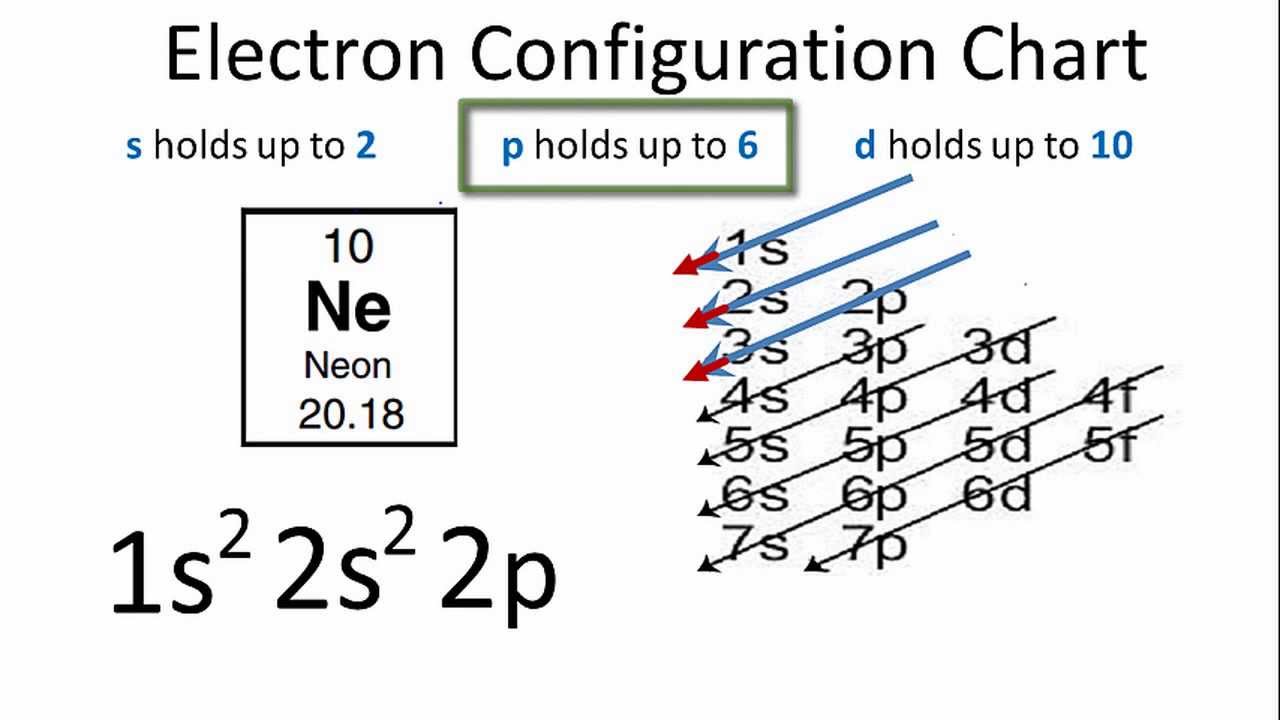
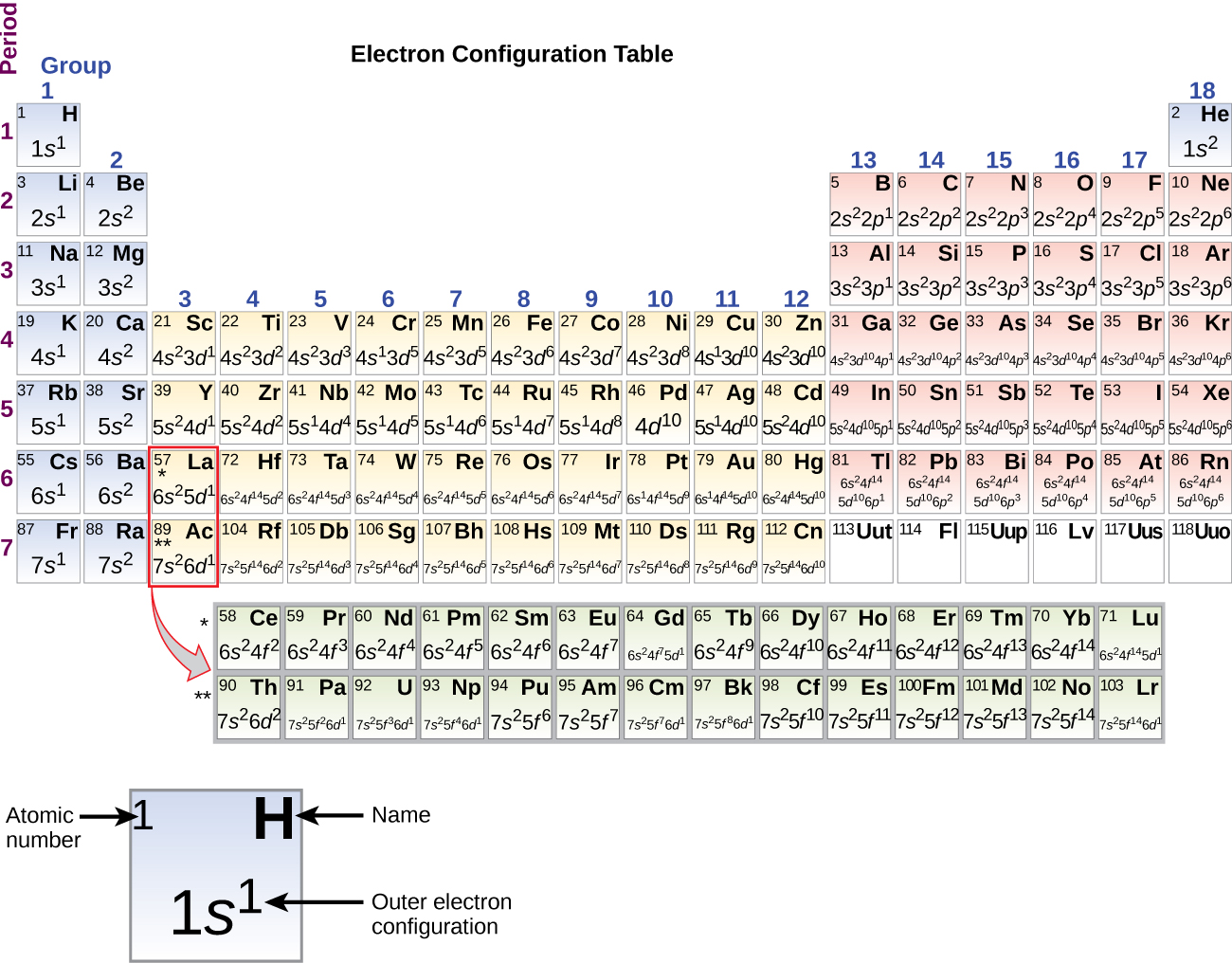
Oxygen Orbital diagram, Electron configuration, and Valence electron Oxygen is situated in Group 16th and has an atomic number of 8. The first shell of Oxygen has 2 electrons and the outer shell or valence shell of oxygen has 6 electrons, hence, the number of valence electrons in the Oxygen atom is 6. The orbital diagram for Oxygen is drawn by following three principles - the Aufbau principle, Hund's ... Neon(Ne) electron configuration and orbital diagram The electron configuration of all the elements can be done through the orbital diagrams. Electron configuration of neon through orbital Atomic energy shells are subdivided into sub-energy levels. These sub-energy levels are also called orbital. The most probable region of electron rotation around the nucleus is called the orbital.
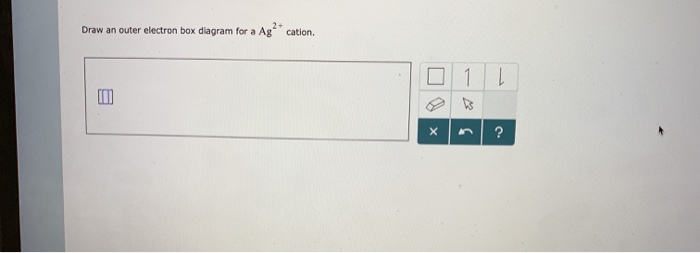
Question : Draw an outer electron box diagram for a Fe+3 cation. - Chegg Draw an outer electron box diagram for a Fe+3 cation. Question: Draw an outer electron box diagram for a Fe+3 cation. This problem has been solved! See the answer Draw an outer electron box diagram for a Fe+3 cation. Expert Answer 100% (11 ratings) If you have any qu … View the full answer Previous question Next question
Outer electron box diagram
What is the electron configuration for Zn2+? | Socratic The electron configuration of Zn2+ is 1s22s22p63s23p63d10. Zinc is a d-block element, also known as a transition element. For the d-block elements, the outermost s-sublevel has higher energy than the d-sublevel, which is contrary to what the Aufbau diagram indicates. Argon Orbital diagram, Electron configuration, and Valence electrons The orbital diagram simply represents the arrangement of electrons in the different orbitals of an atom, it uses an arrow to represent the electrons, every orbital (one box) contains a maximum of 2 electrons. There are three rules followed for constructing the orbital diagram for an atom. (1). Bohr Diagrams of Atoms and Ions - Chemistry LibreTexts Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is ...
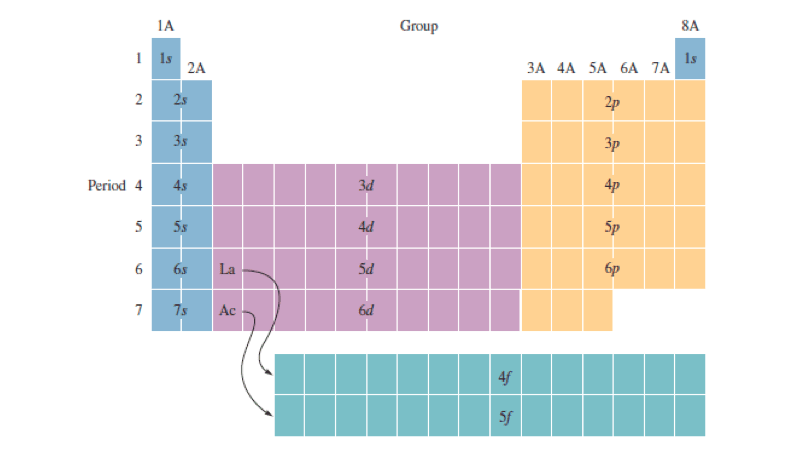
Outer electron box diagram. Electron Configuration of Transition Metals - Chemistry LibreTexts In the first row of the transition metals, the ten elements that can be found are: Scandium (Sc), Titanium (Ti), Vanadium (V), Chromium (Cr), Manganese (Mn), Iron (Fe), Cobalt (Co), Nickel (Ni), Copper (Cu), and Zinc (Zn). Below is a table of the oxidation states that the transition metals can or cannot form. 3.1: Electron Configurations - Chemistry LibreTexts The electron configuration and the orbital diagram are: Following hydrogen is the noble gas helium, which has an atomic number of 2. The helium atom contains two protons and two electrons. The first electron has the same four quantum numbers as the hydrogen atom electron ( n = 1, l = 0, ml = 0, m s = + 1 2 ). Potassium (K) Orbital diagram, Electron configuration, and Valence ... The electron configuration for potassium ion (K +) is 1s 2 2s 2 2p 6 3s 2 3p 6. The number of valence electrons available for the Potassium atom is 1. Potassium is situated in Group 1st and has an atomic number of 19. The first shell of Potassium has 2 electrons and the outer shell or valence shell of Potassium has only 1 electron, hence, the ... Ultrastrong coupling between light and matter | Nature ... Jan 08, 2019 · Light–matter coupling with strength comparable to the bare transition frequencies of the system is called ultrastrong. This Review surveys how experiments have realized ultrastrong coupling in ...
Is the order of orientation of electron box diagrams meaningful or ... This sort of diagram is for representing the quantum numbers of the electrons in the system. The 3d boxes mean the quantum numbers n=3, l=2 with ml =-2,-1,0,+1,+2 for the 5 boxes. If a box has an up and a down arrow in it, this means there are 2 electrons with those quantum numbers, one with s=1/2, and one with 2=-1/2. 3.4: Electronic Structure of Atoms (Electron Configurations) For orbital diagrams, this means two arrows go in each box (representing two electrons in each orbital) and the arrows must point in opposite directions (representing paired spins). The electron configuration and orbital diagram of helium are: The n = 1 shell is completely filled in a helium atom. 40 Electron Configurations, Orbital Box Notation (M7Q7) - Unizin The electron configuration and orbital diagram of helium are: The n = 1 shell is completely filled in a helium atom. The next atom is the alkali metal lithium with an atomic number of 3. The first two electrons in lithium fill the 1 s orbital and have the same sets of four quantum numbers as the two electrons in helium. Electron Box Diagrams - YouTube Electron Box Diagrams
Orbital diagram - How to draw, Examples, Rules, Filling order - Topblogtenz The orbital diagram simply represents the arrangement of electrons in the different orbitals of an atom, it uses an arrow to represent the electrons, every orbital (one box) contains a maximum of 2 electrons. There are three rules followed for constructing the orbital diagram. The three rules are - (a). Aufbau's rule (b). Hund's rule (c). History of the camera - Wikipedia The forerunner to the photographic camera was the camera obscura.Camera obscura (Latin for "dark room") is the natural optical phenomenon that occurs when an image of a scene at the other side of a screen (or for instance a wall) is projected through a small hole in that screen and forms an inverted image (left to right and upside down) on a surface opposite to the opening. Aleks Drawing a box diagram of the electron configuration of an atom ... Aleks Drawing a box diagram of the electron configuration of an atom - YouTube AboutPressCopyrightContact usCreatorsAdvertiseDevelopersTermsPrivacyPolicy & SafetyHow YouTube worksTest new... Cathode-ray tube - Wikipedia A CRT is a glass envelope which is deep (i.e., long from front screen face to rear end), heavy, and fragile. The interior is evacuated to 0.01 pascals (1 × 10 −7 atm) to 0.1 micropascals (1 × 10 −12 atm) or less, to facilitate the free flight of electrons from the gun(s) to the tube's face without scattering due to collisions with air molecules.
Draw an outer electron box diagram for a Nb^{3+} cation. - Study.com Draw an outer electron box diagram for a {eq}Nb^ {3+} {/eq} cation. D - block elements Generally, the electronic configuration of d block elements is {eq} ( n - 1 ) d ^ {1-10} ns^ {1-2} {/eq} (...
Chromium(Cr) electron configuration and orbital diagram - Valenceelectrons The electron configuration of all the elements can be done through the orbital diagram. Electron configuration of chromium through orbital Atomic energy shells are subdivided into sub-energy levels. These sub-energy levels are also called orbital. The most probable region of electron rotation around the nucleus is called the orbital.
Ch. 6: Cellular Respiration Flashcards | Quizlet Draw a block diagram of a feedback system designed to control the amount of HIV viruses in an infected person. The plant input variables are the amount of RTIs and PIs dispensed. Show blocks representing the controller, the system under control, and the transducers.
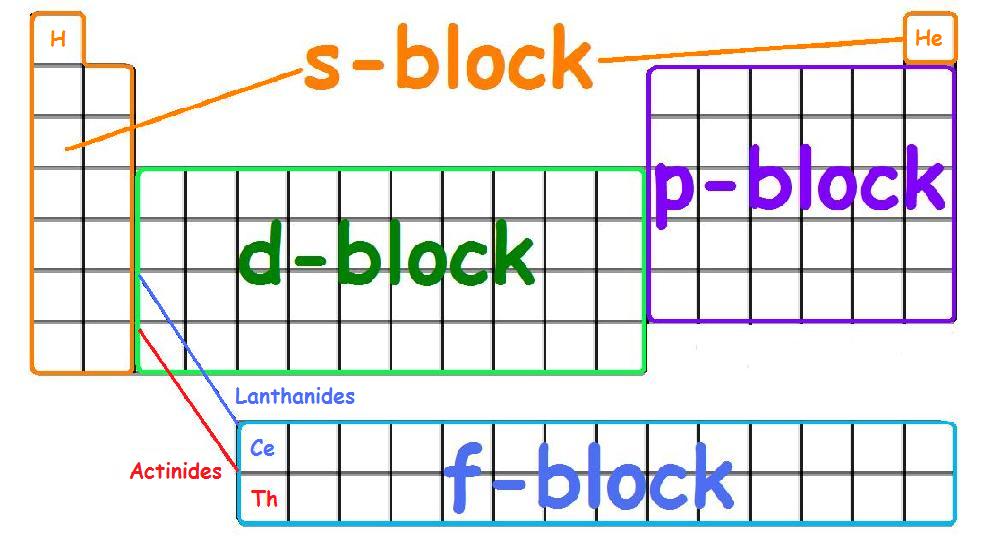
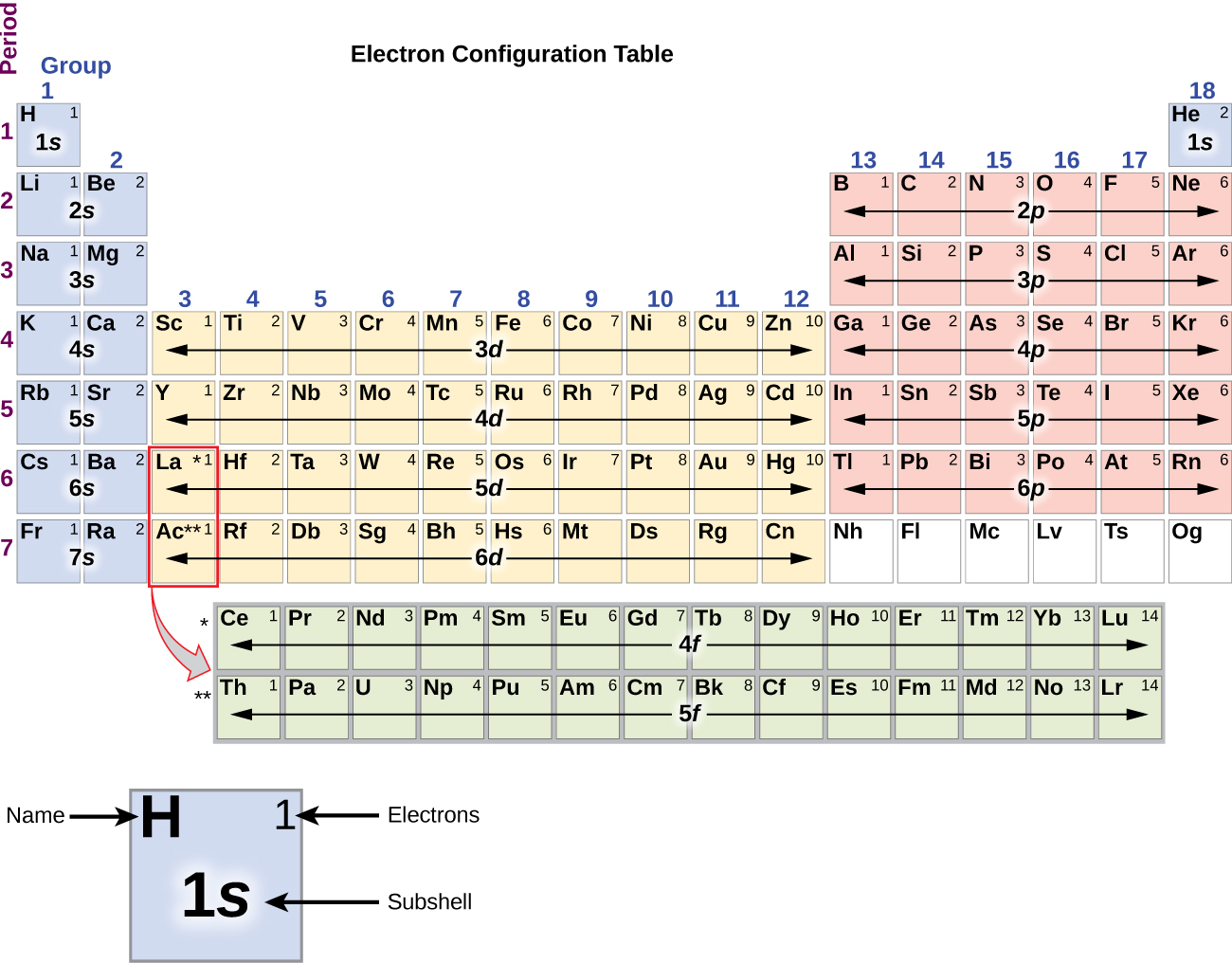
Electron Configuration - Detailed Explanation, Filling of orbital ... Electron Configuration -The Electron Configuration of an Element Describes how Electrons are Distributed in their Atomic Orbitals. In Electronic Configuration electrons are arranged in various shells, Subshell and Orbital by following certain rules. To Learn how to Write Electronic Configurations, Detailed Explanation, Filling of orbital with FAQs, Visit BYJU'S for detailed explanation.
Electron Configurations: Orbital Box Diagrams - YouTube 59,313 views May 11, 2017 675 Dislike Share JFR Science 13K subscribers UPDATE: Thanks to Tessa M., and Spencer K., who pointed out some glaring errors... Mr. Key introduces another representation...
Dynamical system - Wikipedia In mathematics, a dynamical system is a system in which a function describes the time dependence of a point in an ambient space.Examples include the mathematical models that describe the swinging of a clock pendulum, the flow of water in a pipe, the random motion of particles in the air, and the number of fish each springtime in a lake.
Electron Configurations of Ions - UCalgary Chem Textbook Remember electrons are negatively charged, so ions with a positive charge have lost an electron. For main group elements, the last orbital gains or loses the electron. For transition metals, the last s orbital loses an electron before the d orbitals. (a) Na: 1s 2 2s 2 2p 6 3s 1. Sodium cation loses one electron, so Na +: 1s 2 2s 2 2p 6 3s 1 ...
Rhodium(Rh) electron configuration and orbital diagram - Valenceelectrons Rhodium (Rh) electron configuration and orbital diagram Rhodium (Rh) electron configuration and orbital diagram Rhodium is the 45th element in the periodic table and its symbol is 'Rh'. The total number of electrons in rhodium is forty-five. These electrons are arranged according to specific rules of different orbits.
Question : Draw an outer electron box diagram for a Rh cation. Chemistry questions and answers Draw an outer electron box diagram for a Rh cation. Question: Draw an outer electron box diagram for a Rh cation. This problem has been solved! You'll get a detailed solution from a subject matter expert that helps you learn core concepts. See Answer Show transcribed image text Expert Answer 100% (9 ratings)
Electron Configurations and Orbital Box Diagrams An orbital box diagram can be written as well. Boxes, or horizontal lines represent the orbitals, arrows represent the electrons, and if an orbital is full, the electrons must be of opposite spin-one arrow pointing up and the other one pointing down. The orbital box diagrams are listed for the first 20 elements in the figure below.
View 18'th Dynasty Mummies from the Theban Royal Necropolis Her outer coffin's trough lay atop the pile of pottery vessels at the end of the room, and its lid lay across the chamber, resting on some bedsteads and the lid and side panel of her sarcophagus. (See KV 46 diagram, outer coffins highlighted in red.) The positions of the outer coffin lid and basin could be explained as the work of thieves, but ...
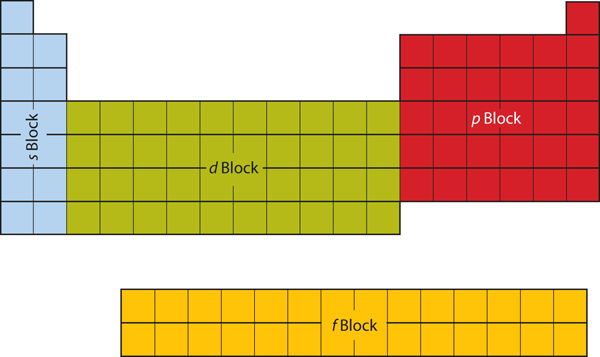
chem 5 Flashcards | Quizlet Study with Quizlet and memorize flashcards containing terms like Mendeleev's original 1870 periodic table illustrated that similar chemical and physical properties recur periodically when the 65 known elements at the time were arranged by ______., The electron configuration of an element describes _____., Which of the following features arise when depicting and populating an orbital energy ...
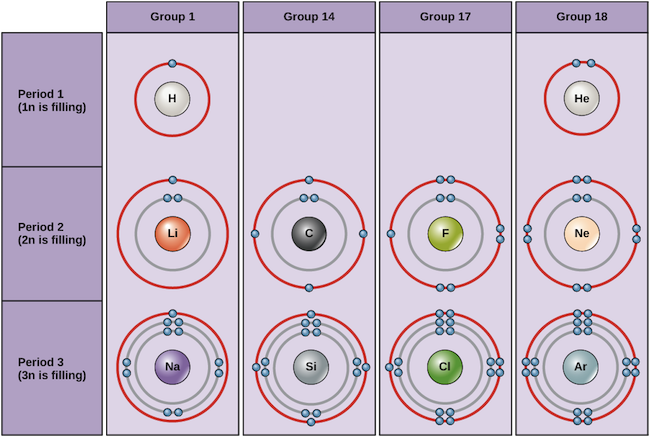
How to draw electron configuration diagrams - RSC Education An electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Electrons are represented by dots or crosses and are positioned in energy levels, or 'shells', around the central nucleus. This is sometimes called the Bohr, or the 'solar system', model. Download this
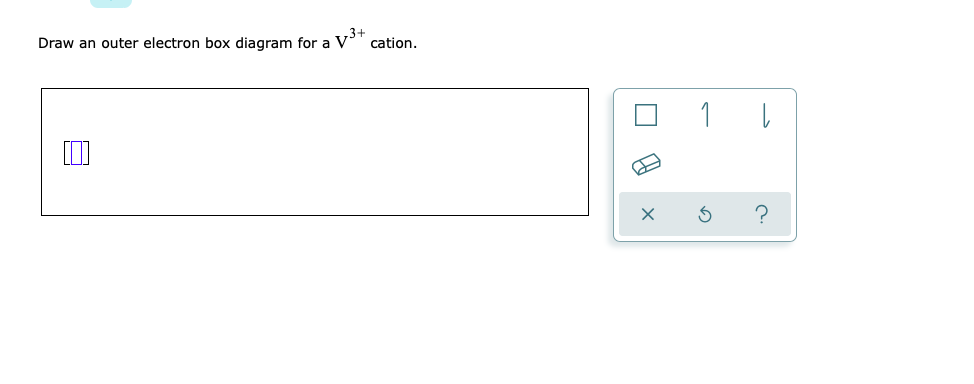
Vanadium(V) electron configuration and orbital diagram - Valenceelectrons The electron configuration of all the elements can be done through the orbital diagram. Electron configuration of vanadium through orbital Atomic energy shells are subdivided into sub-energy levels. These sub-energy levels are also called orbital. The most probable region of electron rotation around the nucleus is called the orbital.
Nitrogen(N) electron configuration and orbital diagram - Valenceelectrons Nitrogen (N) electron configuration and orbital diagram Nitrogen (N) electron configuration and orbital diagram Nitrogen is the 7th element in the periodic table and its symbol is 'N'. The total number of electrons in nitrogen is seven. These electrons are arranged according to specific rules of different orbits.
Orbital Diagram of All Elements (Diagrams given Inside) Orbital diagrams (Orbital box diagrams) of all elements are mentioned in the chart given below. Free Gift for you: Interactive Periodic Table Let me tell you how this Interactive Periodic Table will help you in your studies. 1). You can effortlessly find every single detail about the elements from this single Interactive Periodic table. 2).
Bohr Diagrams of Atoms and Ions - Chemistry LibreTexts Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is ...
Argon Orbital diagram, Electron configuration, and Valence electrons The orbital diagram simply represents the arrangement of electrons in the different orbitals of an atom, it uses an arrow to represent the electrons, every orbital (one box) contains a maximum of 2 electrons. There are three rules followed for constructing the orbital diagram for an atom. (1).
What is the electron configuration for Zn2+? | Socratic The electron configuration of Zn2+ is 1s22s22p63s23p63d10. Zinc is a d-block element, also known as a transition element. For the d-block elements, the outermost s-sublevel has higher energy than the d-sublevel, which is contrary to what the Aufbau diagram indicates.

Which of the following outer electron configurations would you expect to belong to a reactive metal?


















:max_bytes(150000):strip_icc()/800px-Orbital_representation_diagram.svg-589bd6285f9b58819cfd8460.png)

0 Response to "41 outer electron box diagram"
Post a Comment