40 orbital diagram for argon
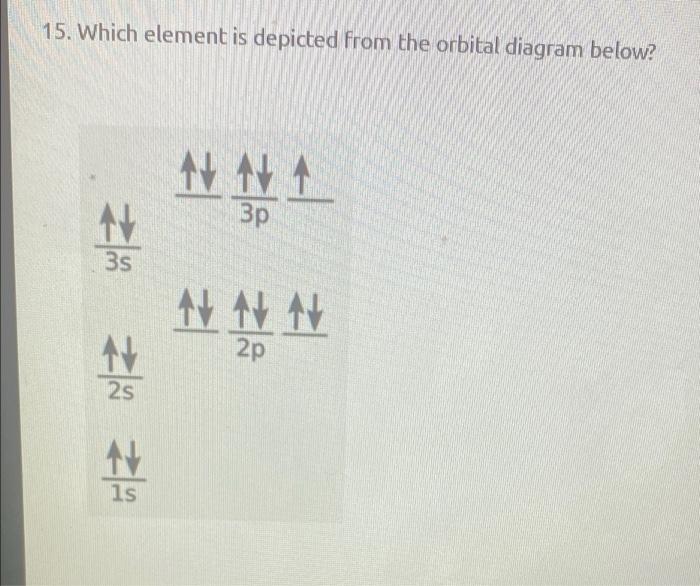
How to Write the Atomic Orbital Diagram for Argon (Ar) To write the orbital diagram for the Argon (Ar) first we need to write the electron configuration for just Ar. To do that we need to find the number of electrons for the Ar atom (there are 18... Orbital Diagram Practice | Chemistry Quiz - Quizizz What is incorrect about this orbital diagram? answer choices Both arrows in the 2p box should be pointing up There is nothing incorrect with this diagram In the 2p box there should only be 1 electron in the first 2p box and one in the 2nd 2p box All the arrows should be pointing up. Question 8 300 seconds Q. What is this element?
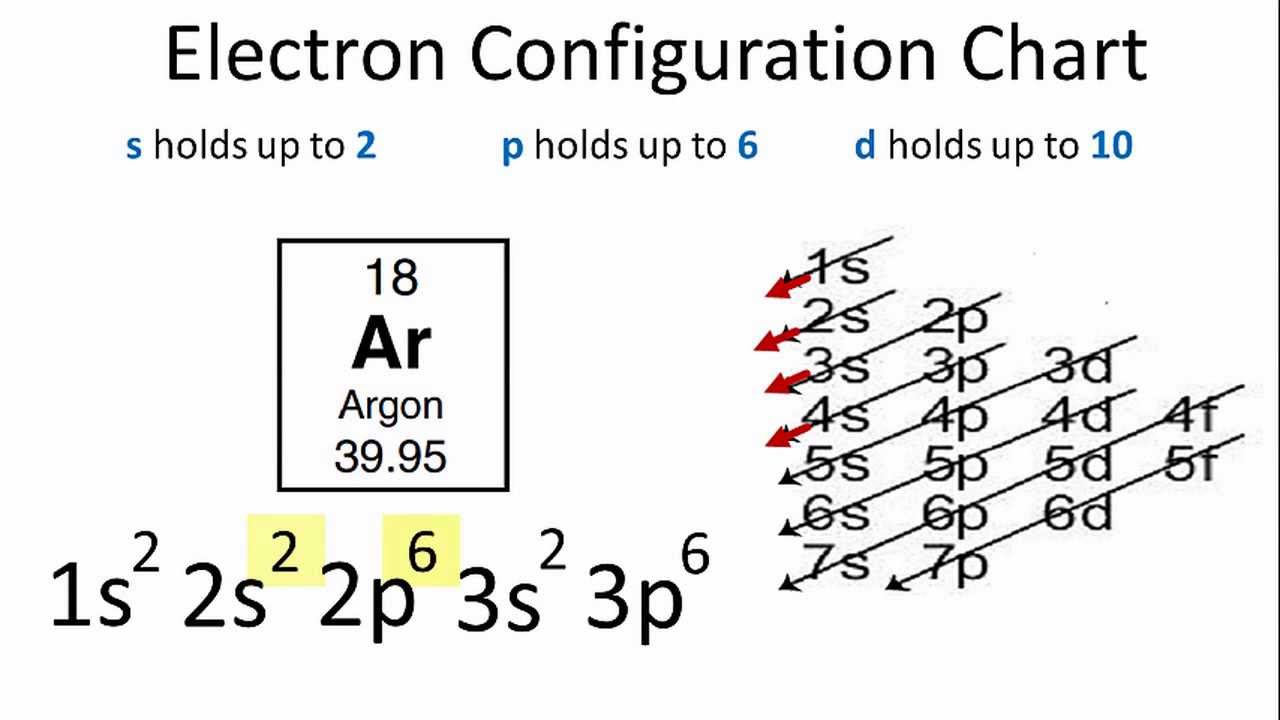
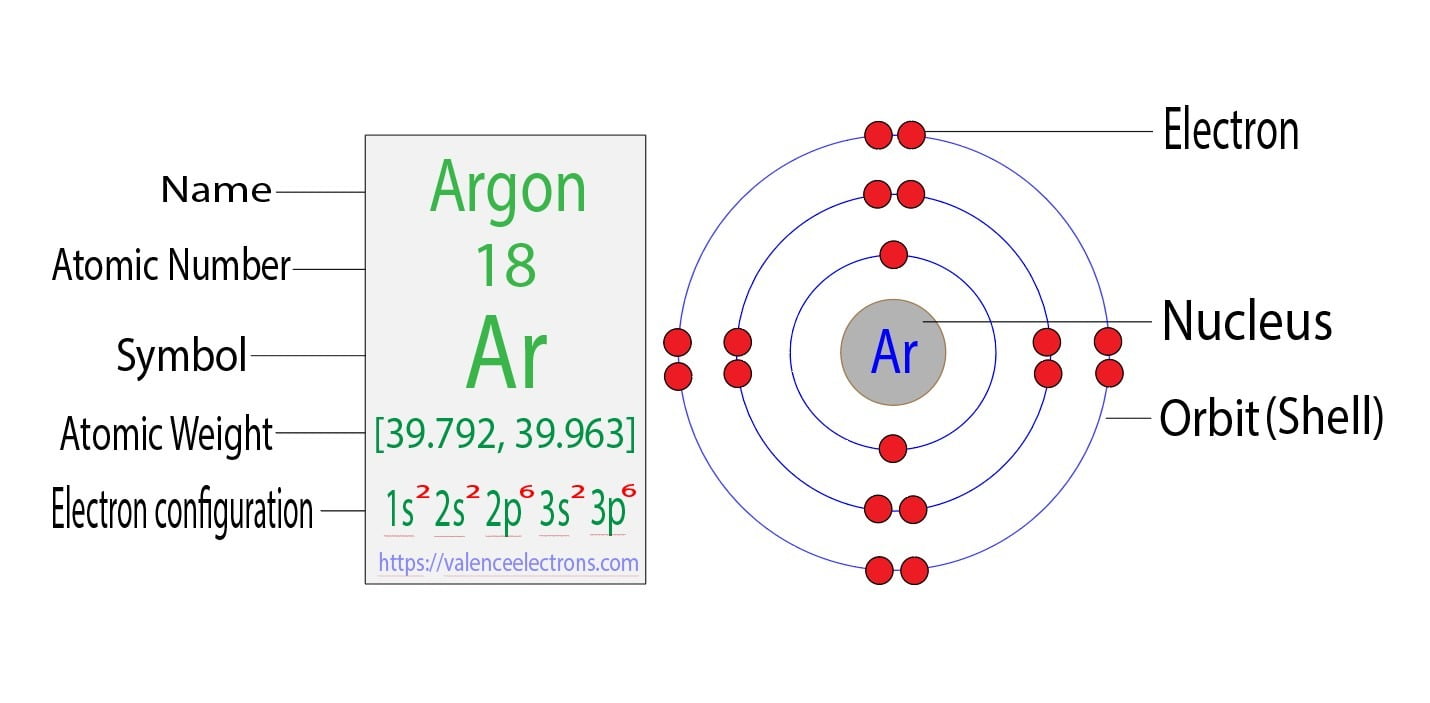
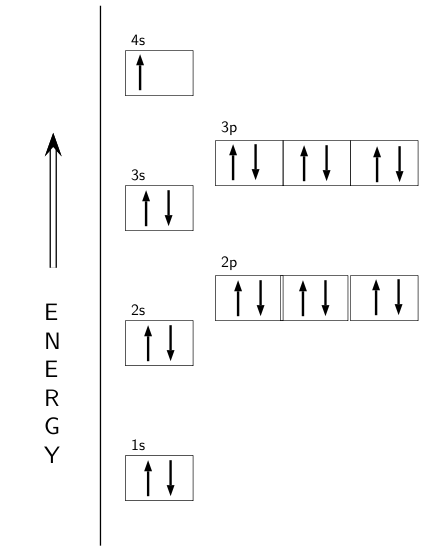
Electron configuration for Argon (element 18). Orbital diagram Density: 0.00166 g/cm 3 . Electronic configuration of the Argon atom: 1s 2 2s 2 2p 6 3s 2 3p 6. Reduced electronic configuration Ar: [Ne] 3s 2 3p 6. Below is the electronic diagram of the Argon atom Distribution of electrons over energy levels in the Ar atom. 1-st level (K): 2. 2-st level (L): 8. 3-st level (M): 8.

Orbital diagram for argon
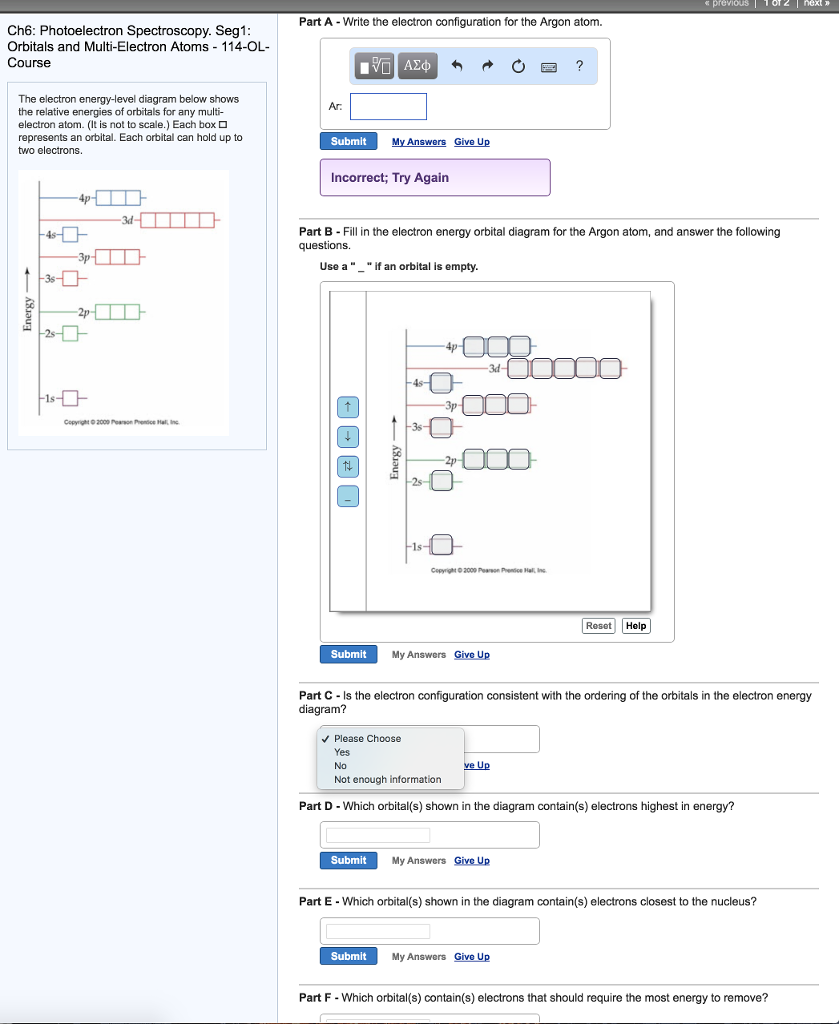
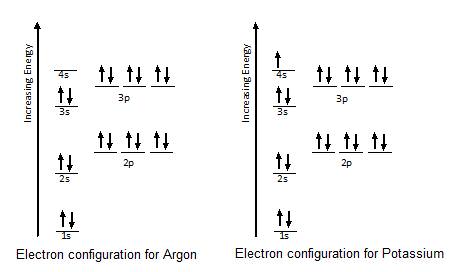
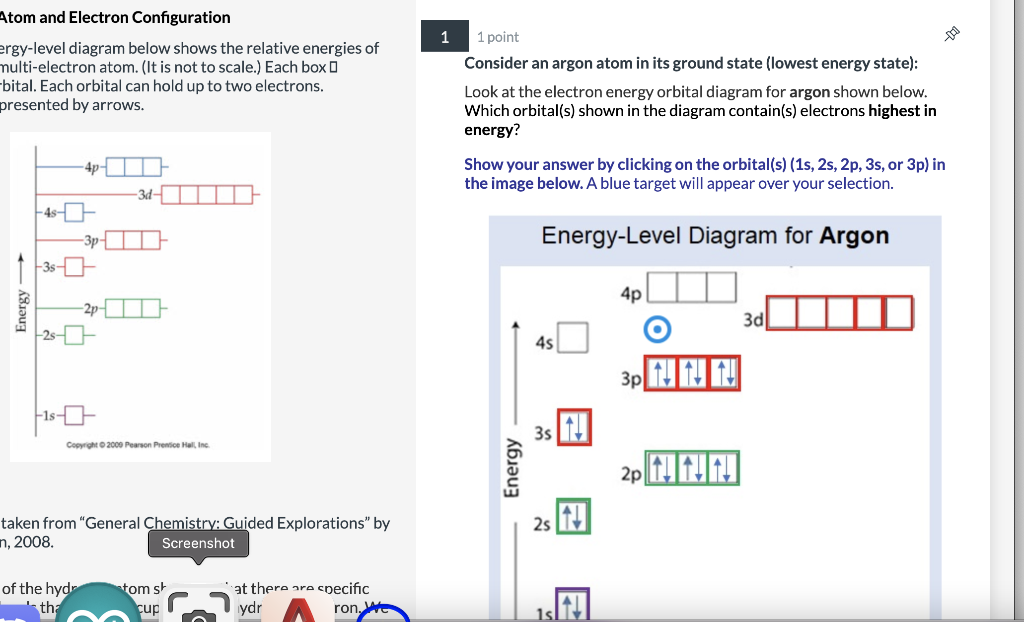
3.7 Orbital Diagrams (Chemistry) Flashcards | Quizlet Identify the element from the electron configuration: [Ar]4s^2 3d^2. Ti. First, we see that the electron configuration is made of the core electrons (Ar) and the valence electrons (4s23d2). Since there are additional electrons after the nobel gas, argon, we know that this element must be from period 4. Radon(Rn) electron configuration and orbital diagram - Valenceelectrons To write the orbital diagram of radon (Rn), you have to do the electron configuration of radon. Which has been discussed in detail above. 1s is the closest and lowest energy orbital to the nucleus. Therefore, the electron will first enter the 1s orbital. Solved Fill in the orbital energy diagram for argon. 3p 3s E | Chegg.com Expert Answer. 100% (14 ratings) Argon is a nobel gas element, it has …. View the full answer. Transcribed image text: Fill in the orbital energy diagram for argon. 3p 3s E 2p 2s བ ། 1s.
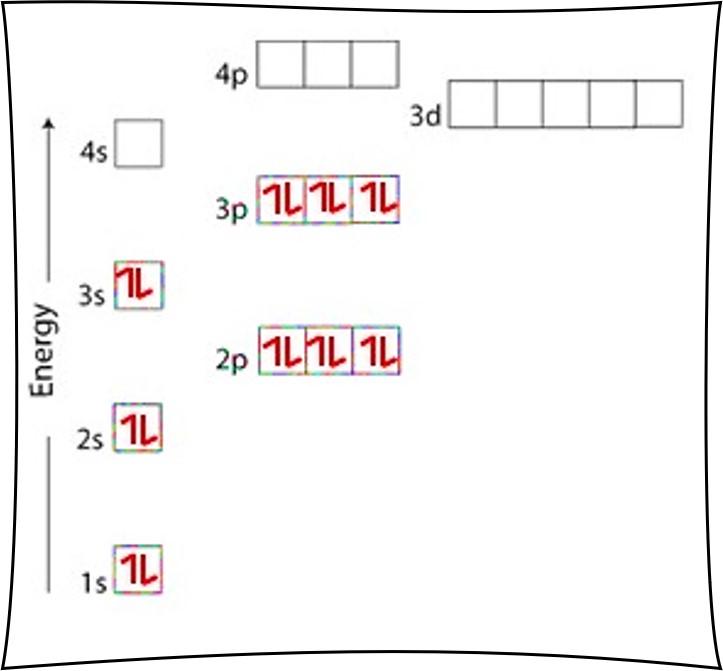
Orbital diagram for argon. Solved Part A Write an orbital diagram for argon Drag the | Chegg.com Part A Write an orbital diagram for argon Drag the appropriate labels to their respective targets. Labels can be used once, more than once, or not at all. Orbital Diagram of Argon - YouTube This video shows how to create an orbital diagram of an atom from its electronic configuration Argon Electron Configuration - Learnool Learn how to draw: Argon Bohr Model #4 From its Orbital Diagram The above orbital diagram shows that the 1s subshell has 2 electrons, the 2s subshell has 2 electrons, the 2p subshell has 6 electrons, the 3s subshell has 2 electrons, and the 3p subshell has 6 electrons. So the electron configuration of argon will be 1s 2 2s 2 2p 6 3s 2 3p 6. Krypton Orbital Diagram This is one of the happy elements and has an electron configuration of The other inert gases including argon and xenon also have full outer shells with eight.In writing the electron configuration for Argon the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for Argon go in the 2s orbital.
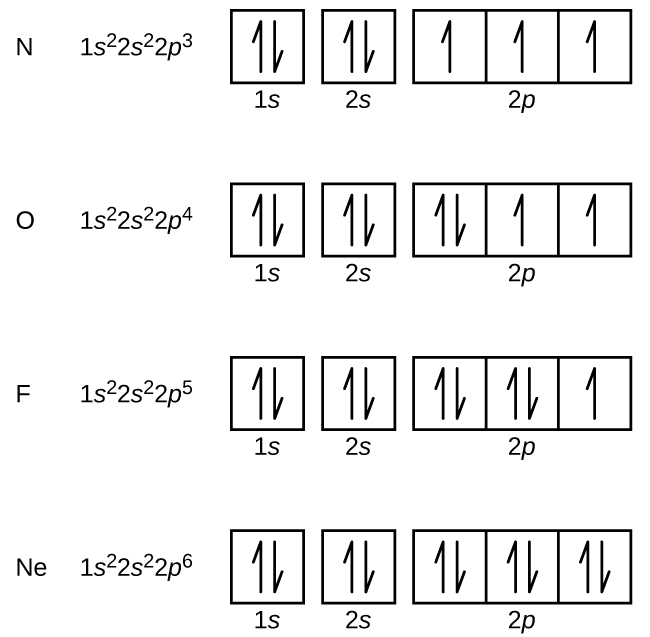
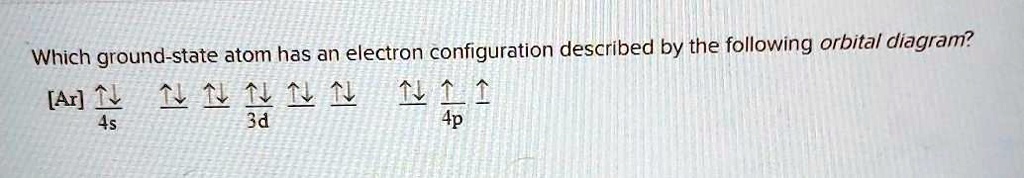
Orbital Diagram of All Elements (Diagrams given Inside) Orbital diagrams (Orbital box diagrams) of all elements are mentioned in the chart given below. Free Gift for you: Interactive Periodic Table Let me tell you how this Interactive Periodic Table will help you in your studies. 1). You can effortlessly find every single detail about the elements from this single Interactive Periodic table. 2). Give the symbol of the atom with the orbital diagram beyond argon. Okay, so for this first one, we'll go from our orbital diagram to an electron configuration, they said it's after Oregon. So for us to three D eight. So you can either count over on the periodic table and see that that's nickel Or you can see that you have 18 close to of course a is 28 electrons which matches up with nickel. Okay, the next one we've got are gone for us to three D five. Orbital Diagrams, Electron Configuration, Valence electrons - Quizlet Start studying Orbital Diagrams, Electron Configuration, Valence electrons. Learn vocabulary, terms, and more with flashcards, games, and other study tools. Orbital diagrams | Chemistry Quiz - Quizizz Both arrows in the 2p box should be pointing up. There is nothing incorrect with this diagram. In the 2p box there should only be 1 electron in the first 2p box and one in the 2nd 2p box. All the arrows should be pointing up. Question 5. 120 seconds. Q. Electrons occupy orbitals of lowest energy first is part of what electron configuration rule?
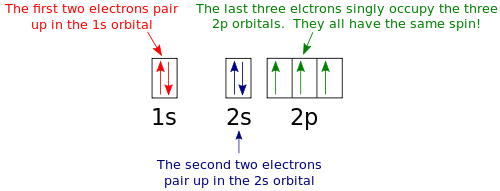
Argon(Ar) electron configuration and orbital diagram - Valenceelectrons Argon orbital diagram According to Hund's principle, the first electron will enter in the clockwise direction and the next electron will enter the 1s orbital in the anti-clockwise direction. The 1s orbital is now filled with two electrons. The next two electrons will enter the 2s orbital just like the 1s orbital. Argon Orbital Diagram - Learnool Argon Orbital Diagram The orbital diagram of argon shows that the 1s subshell has 2 electrons, the 2s subshell has 2 electrons, the 2p subshell has 6 electrons, the 3s subshell has 2 electrons, and the 3p subshell has 6 electrons. Steps Here's how you can draw the orbital diagram of argon step by step. Step #1: find electrons of argon What is the orbital diagram of Argon? - Answers Argon has completely filled M shell (or 3p orbital) What is an orbital diagram? An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by... orbital diagram for argon Beryllium diagram element representation ... We have 9 Pictures about Show The Orbital Filling Diagram For Br Bromine like Argon, atomic structure - Stock Image C018/3699 - Science Photo Library, Electron Configuration And Orbital Diagram For Aluminum - Diagram Media and also Science Presentation by Luis Miguel Bulosan. Here you go: Show The Orbital Filling Diagram For Br Bromine
Electron Configuration for Argon (Ar) - UMD When we write the configuration we'll put all 18 electrons in orbitals around the nucleus of the Argon atom. In writing the electron configuration for Argon the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for Argon go in the 2s orbital. The next six electrons will go in the 2p orbital.
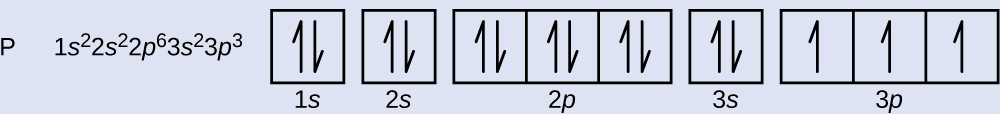
Write the complete orbital configuration for argon. Answer and Explanation: 1 Argon has the atomic number 18 and thus, the full electronic configuration of argon is [Ar] = 1s22s22p63s23p6 [ A r] = 1 s 2 2 s 2 2 p 6 3 s 2 3 p 6 . Here, the...
Argon - Element information, properties and uses | Periodic Table Thermal conductivity of argon at room temperature (300 K) is 17.72 mW m-1 K-1 (milliWatts per metre per degree) whereas for air it is 26 mW m-1 K-1. For the same reason argon is used to inflate diving suits. Old documents and other things that are susceptible to oxidation can be protected by being stored in an atmosphere of argon.
How to Do Orbital Diagrams | Sciencing Because the configuration of argon is: [Ar] = 1s 2 2s 2 2p 6 3s 2 3p 6 You can use this with any elements apart from hydrogen and helium. Orbital Diagrams Orbital diagrams are like the configuration notation just introduced, except with the spins of electrons indicated.
Orbital Diagram For Carbon (C) | Carbon Electron Configuration Carbon Electron Configuration is an element of the periodic table and its atomic number is 6. How many users might wonder what exactly will be the problem with that? The element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. The symbol of carbon is written as 6.
Argon Orbital diagram, Electron configuration, and Valence electrons Orbital diagram for Argon The orbital diagram simply represents the arrangement of electrons in the different orbitals of an atom, it uses an arrow to represent the electrons, every orbital (one box) contains a maximum of 2 electrons. There are three rules followed for constructing the orbital diagram for an atom. (1).
Argon Orbital Energy Diagram MP3 Download (2.86 MB ... Get Argon Orbital Energy Diagram MP3 Download (2.86 MB) on Navidbiglarimusic, Quick and Easy - NAVID BIGLARI MUSIC How To Write The Atomic Orbital Diagram For Argon (Ar) 02:05 min 320 kbps 2.86 MB Downlod Now. Orbital Diagram Of Argon 07:26 min 320 ...
How many unpaired electrons does argon have? - Quora Answer (1 of 5): It is a partial question and the the two answers already given are partial answers. Argon's atomic number is 18 and there are 18 electrons in each atom. Under appropriate physical conditions one can make all the 18 electrons unpaired or 16, 14, 12, 10, 8, 6, 4, 2 electrons unpair...
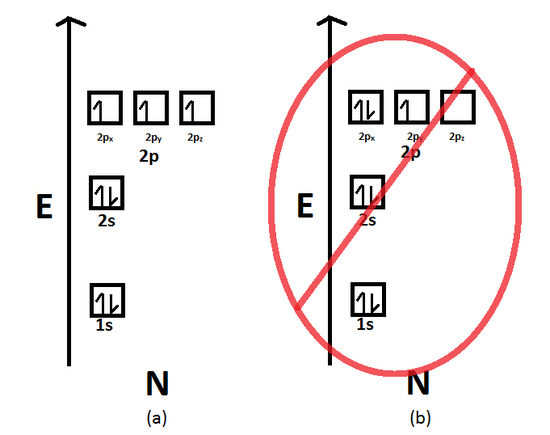
What is the orbital diagram? - NSN search Orbital diagrams are pictorial descriptions of the electrons in an atom. Three rules are useful in forming orbital diagrams. According to the Auf Bau Principle, each electron occupies the lowest energy orbital. The Pauli Exclusion Principle says that only two electrons can fit into an single orbital.
Orbital diagram - How to draw, Examples, Rules, Filling order - Topblogtenz The orbital diagram simply represents the arrangement of electrons in the different orbitals of an atom, it uses an arrow to represent the electrons, every orbital (one box) contains a maximum of 2 electrons. There are three rules followed for constructing the orbital diagram. The three rules are - (a). Aufbau's rule (b). Hund's rule (c).
Solved Fill in the orbital energy diagram for argon. 3p 3s E | Chegg.com Expert Answer. 100% (14 ratings) Argon is a nobel gas element, it has …. View the full answer. Transcribed image text: Fill in the orbital energy diagram for argon. 3p 3s E 2p 2s བ ། 1s.
Radon(Rn) electron configuration and orbital diagram - Valenceelectrons To write the orbital diagram of radon (Rn), you have to do the electron configuration of radon. Which has been discussed in detail above. 1s is the closest and lowest energy orbital to the nucleus. Therefore, the electron will first enter the 1s orbital.
3.7 Orbital Diagrams (Chemistry) Flashcards | Quizlet Identify the element from the electron configuration: [Ar]4s^2 3d^2. Ti. First, we see that the electron configuration is made of the core electrons (Ar) and the valence electrons (4s23d2). Since there are additional electrons after the nobel gas, argon, we know that this element must be from period 4.








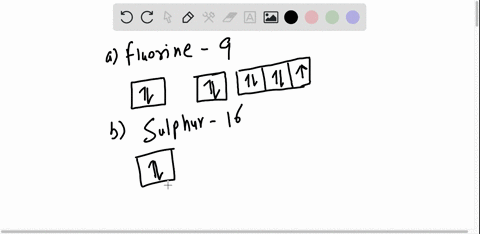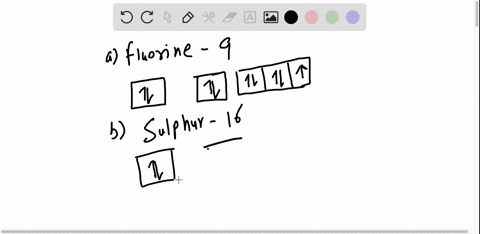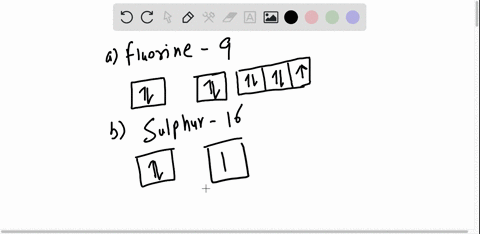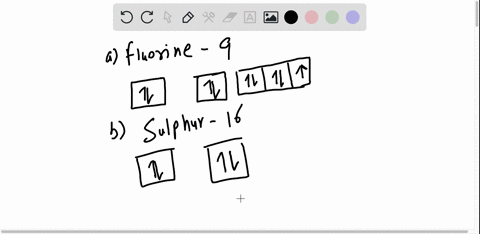

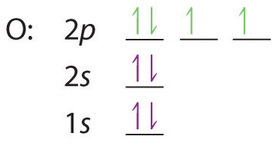












0 Response to "40 orbital diagram for argon"
Post a Comment